Lipid Nanoparticles (LNPs) for Gene Delivery
From formulation development to scalable manufacturing, Curapath supports biotech and pharmaceutical partners with advanced LNP design, targeted delivery strategies and next-generation lipid technologies.
What Are Lipid Nanoparticles (LNPs)?
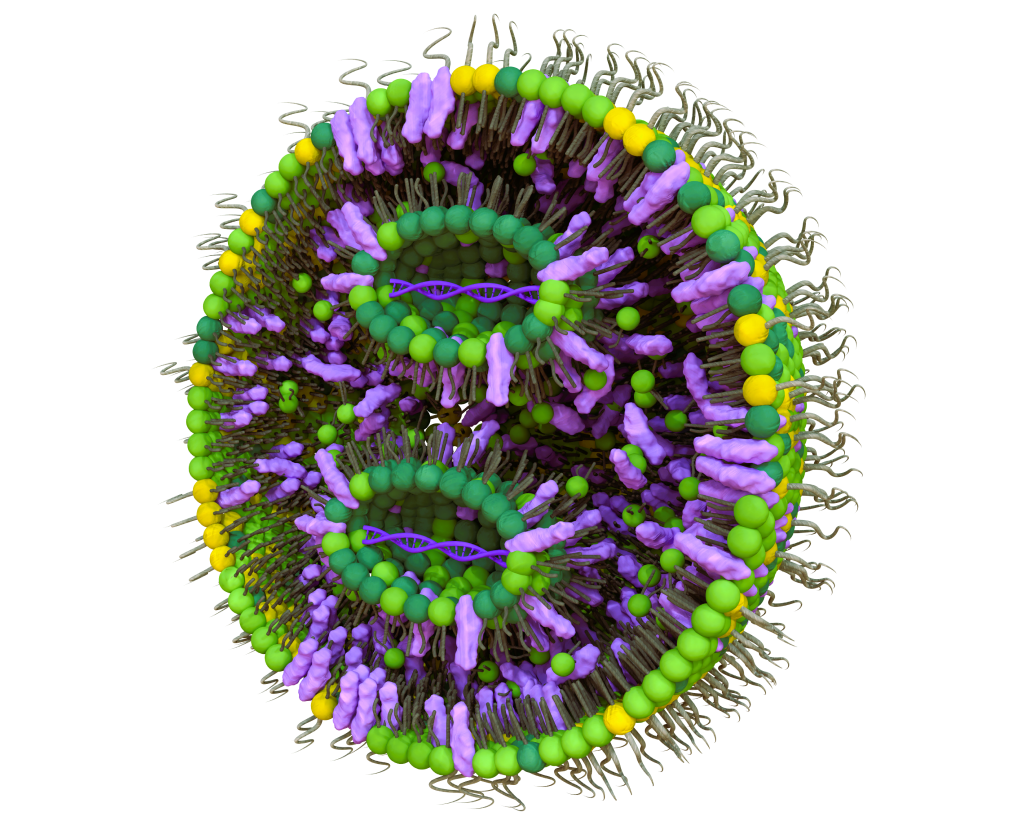
Lipid nanoparticles (LNPs) are advanced non-viral delivery systems designed to encapsulate and transport nucleic acids such as mRNA, siRNA and gene editing payloads into target cells. Their lipid-based structure enables efficient protection of genetic material while facilitating cellular uptake and intracellular delivery.
These nanoparticles play a key role in modern nucleic acid therapeutics by protecting RNA molecules from degradation and improving their stability in biological environments. Once internalized by cells, LNP formulations help release the therapeutic cargo into the cytoplasm, enabling the intended biological activity of the delivered genetic material.
Due to their versatility and scalability, lipid nanoparticles have become a cornerstone technology for RNA therapeutics, including vaccines, gene therapies and emerging genome editing applications.
Applications of Lipid Nanoparticles (LNPs)
Lipid nanoparticles (LNPs) have become a key platform for the delivery of nucleic acid therapeutics across a wide range of applications. Their ability to efficiently encapsulate and transport genetic material has enabled major advances in RNA-based medicines, gene therapy and emerging genome editing technologies.
mRNA Therapeutics and Vaccines
LNPs play a central role in mRNA-based therapeutics, enabling the delivery of messenger RNA into cells for protein expression. This technology has been widely used in vaccine development and continues to expand into new therapeutic areas.Advanced LNP design allows fine-tuning of delivery efficiency and expression profiles depending on the therapeutic objective..
siRNA and Gene Silencing
LNPs enable the targeted delivery of small interfering RNA (siRNA), allowing selective gene silencing. This approach is particularly relevant for treating genetic disorders and diseases driven by specific gene expression.
Optimized lipid composition and particle design are key to achieving effective and tissue-specific gene silencing.
Gene Editing (CRISPR and Beyond)
LNP systems are increasingly used to deliver gene editing tools such as CRISPR-Cas systems. Their non-viral nature provides a safer alternative for transient delivery and controlled genome modification.
LNP-based delivery enables transient and controlled exposure to gene editing systems, reducing long-term safety concerns.d.
Oncology and Targeted Therapies
LNPs are being explored for cancer treatment through the delivery of RNA therapeutics and immunomodulatory agents. Advanced formulations enable targeted delivery to specific tissues or cell types.
Next-generation LNP platforms incorporate targeting strategies to improve biodistribution and enhance therapeutic efficacy in oncology applications.

Designing LNP systems for different applications requires a deep understanding of lipid composition, formulation parameters and delivery challenges. At Curapath, we support partners in developing tailored LNP solutions from early formulation to scalable manufacturing.
Challenges in LNP Development and Curapath’s Approach
Endosomal Escape: the challenge
Efficient endosomal escape remains one of the main limitations of LNP systems. Although nanoparticles are readily internalized by cells through endocytosis, only a small fraction of the nucleic acid cargo is successfully released into the cytoplasm, limiting overall delivery efficiency
Endosomal Escape: our approach
Optimization of ionizable lipid composition and formulation parameters plays a key role in enhancing membrane destabilization and promoting endosomal escape. Fine-tuning pKa values and lipid ratios allows improved intracellular release while maintaining an acceptable safety profile.
Biodistribution and Targeting: the challenge
The biodistribution of LNPs is often biased towards hepatic accumulation, which can limit their applicability for therapies requiring delivery to other tissues or cell types. Achieving controlled and targeted delivery remains a key challenge in LNP design.
Biodistribution and Targeting: our approach
Advanced formulation strategies and lipid engineering enable modulation of biodistribution profiles. By adjusting lipid composition and incorporating targeting elements, LNP systems can be tailored to improve delivery to specific tissues beyond the liver.
Toxicity and Tolerability: the challenge
Certain lipid components, particularly ionizable lipids and PEGylated lipids, can induce immune responses or toxicity. Balancing delivery efficiency with biocompatibility is essential for successful clinical translation.
Toxicity and Tolerability: Our approach
Careful selection and optimization of lipid components, including the use of PEG alternatives and shielding strategies, helps improve tolerability while maintaining performance. Formulation design focuses on minimizing immunogenicity without compromising delivery efficiency.
Stability and Storage: the challenge
LNP formulations can be sensitive to environmental conditions such as temperature and pH, which may affect their stability during storage and distribution. Maintaining structural integrity over time is critical for therapeutic performance.
Stability and Storage: our approach
Stabilization strategies, including optimized formulation design and the use of cryoprotectants or lyophilization processes, are implemented to ensure long-term stability and consistent performance across storage conditions.Also aous Alternative shielding lipids have demonstrated excepcional perfomance and enable Freeze-dried LNP formulations
Scalability and Reproducibility: the challenge
Scaling LNP formulations from laboratory development to GMP manufacturing requires tight control over process parameters. Small variations can significantly impact particle size, encapsulation efficiency and overall performance.
Scalability and Reproducibility: our approach
The implementation of controlled and reproducible manufacturing processes, such as microfluidic mixing, enables consistent LNP production. Process optimization ensures seamless transition from early development to large-scale GMP manufacturing.
End-to-End Lipid Nanoparticle (LNP) Development and Manufacturing Workflow
Raw Materials & Preparation
Lipid nanoparticle (LNP) development begins with the selection and preparation of high-quality raw materials, including lipids, nucleic acids and formulation buffers. Proper handling and preparation of these components are critical to ensure consistency and reproducibility in downstream processes.
At this stage, key parameters such as nucleic acid quality, buffer composition and lipid purity are defined to support efficient LNP formulation and optimal performance.
LNP Formulation

Clinical Development and GMP Manufacturing Services

Lipid Nanoparticle Formulation
LNP formulation involves the precise combination of lipid components and nucleic acids to achieve efficient encapsulation and desired particle characteristics. Critical parameters such as lipid ratios, N/P ratio and formulation conditions are optimized to ensure high encapsulation efficiency and stability.
At Curapath we support formulation development that is tailored to the specific therapeutic application, enabling the design of LNP systems with controlled size, charge and delivery performance.
Commercial supply of polymer and lipid excipients and nanoparticle formulations

Purification
Following nanoparticle formation, purification processes are applied to remove residual solvents, unencapsulated nucleic acids and impurities. Techniques such as tangential flow filtration (TFF) or dialysis are commonly used to ensure product quality and consistency.
Effective purification is essential to improve safety, stability and overall performance of the LNP formulation.
Concentration
LNP concentration and final formulation steps are used to adjust particle concentration and buffer composition according to the intended application. This stage may include the addition of cryoprotectants or stabilizing agents to enhance product stability.
Careful control of formulation parameters ensures that LNP systems maintain their physicochemical properties during storage and handling.


Filtration
Sterile filtration is a critical step in LNP manufacturing, ensuring product sterility and compatibility with clinical and GMP requirements. Filter selection and validation are essential to prevent particle loss or changes in LNP characteristics.
Process optimization focuses on maintaining product integrity while achieving the required sterility standards.
Fill & Finish
Fill and finish operations involve the aseptic filling of LNP formulations into final containers, such as vials or syringes. This stage includes the definition of filling conditions, container selection and handling procedures.
Ensuring product integrity and sterility during fill and finish is essential for clinical and commercial applications.

Curapath Capabilities in Lipid Nanoparticle (LNP) Development

LNP Formulation Development
Design and optimization of LNP formulations tailored to specific nucleic acid therapeutics, including mRNA, siRNA and gene editing applications.
Discover our Formulation development packages
Lipid Exciptients manufacturing
Development and integration of next-generation lipid systems, including alternative ionizable lipids, PEG alternatives and shielding lipids to improve safety and enable repeat dosing.
Discover all components available

Targeted Delivery Strategies
Design of LNP systems with enhanced biodistribution and targeting capabilities to support extrahepatic delivery and advanced therapeutic applications.

Process Development and Scale-Up
Optimization of formulation processes and scale-up strategies to ensure reproducibility and seamless transition to manufacturing

GMP Manufacturing
Production of LNP formulations under GMP conditions, supporting clinical and commercial supply of advanced therapeutics.
LNP Resources
LNP Anaytical methods
Lipid Nanoparticle Formulation Characterization: Bridging the Gap from R&D to GMP Scale through Robust Analytical Excellence
LNP formulation Process
Unlocking the Potential of Lipid Nanoparticles:
A Journey Towards GMP-Compliant Formulation Development
Next Generation LNP
Certest and Curapath have joined forces to develop a novel LNP formulation leveraging the complementary Benefits of their proprietary excipients
Formulation Development Services
Discover Curapath’s Essential, Advanced, and Expert formulation packages for LNP systems, flexible, cost-effective solutions for drug delivery development.
Active Targeting Shielding Lipids
Discover CliCr®-modified shielding lipids designed to enhance functionalization density, maintain particle integrity, and enable targeted delivery
Product Catalog
Explore a vast selection of components for you LNP formulations, from FTO ionizable lipids to PEG-FREE shielding lipids